The revision from ICH E6(R2) to E6(R3) was long overdue. Here’s what changed and what it means in practice.
📍 Context
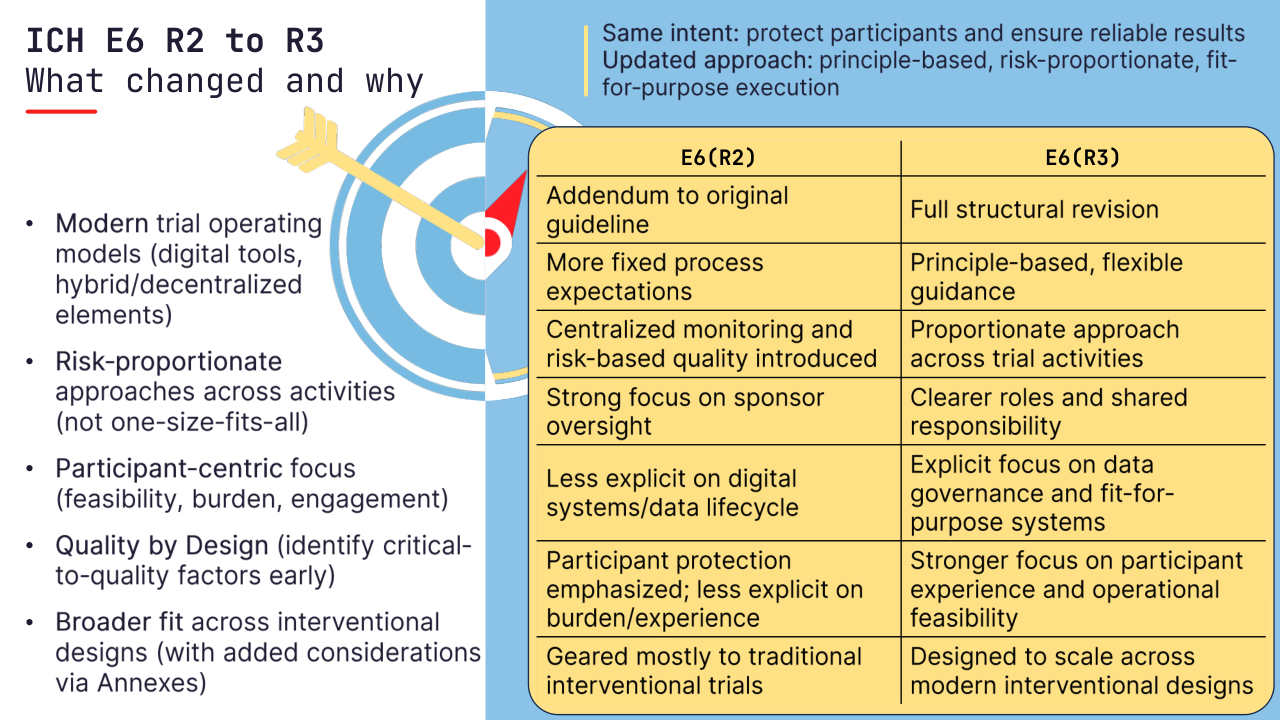
R2 (2016) strengthened oversight: centralized monitoring, risk-based quality management, and clearer sponsor responsibilities. Since then, trials have become more digital and distributed:
• decentralized/hybrid elements;
• real-time data streams from multiple systems;
• higher expectations for transparency and participant experience;
• risk-based approaches becoming the default.
📍 What R3 is really doing
E6(R3) keeps the same intent (participant protection + reliable results), but shifts from process-heavy “how-to” prescriptions to principle-based, risk-proportionate, fit-for-purpose ways to get there.
📍 Five drivers behind the change
Risk proportionality – scale controls to what can impact safety or the reliability of key endpoints.
Data governance – digital systems, data lifecycle, and traceability are now first-class GCP topics.
Quality by design – build quality into design, not only via retrospective QC
Participant focus – reduce burden, improve feasibility and retention.
Broader applicability – designed to better accommodate a wider range of interventional trial designs (incl. decentralized/pragmatic elements, with additional considerations in Annexes).
📍 What this means for data and stats teams (tomorrow morning)
• Focus controls on what can bias the estimator or compromise endpoint reliability – not maximizing SDV
• Make traceability inspection-ready: Protocol → SAP → ADaM → TFLs with reproducible programming and QC evidence
• Treat vendor/digital sources (eCOA, wearables, local labs) as part of your governance model: fit-for-purpose + documented oversight
In E6(R3), the original 13 principles from E6(R2) are consolidated into 11 principles. Two themes become explicit and stronger: risk proportionality and roles and responsibilities and all principles come with more practical explanation.
🔜 Next: Principles 1–3: what changes in ethical framing, consent, and independent review.