Now we move from design principles to making execution feasible and ensuring the results are reliable.
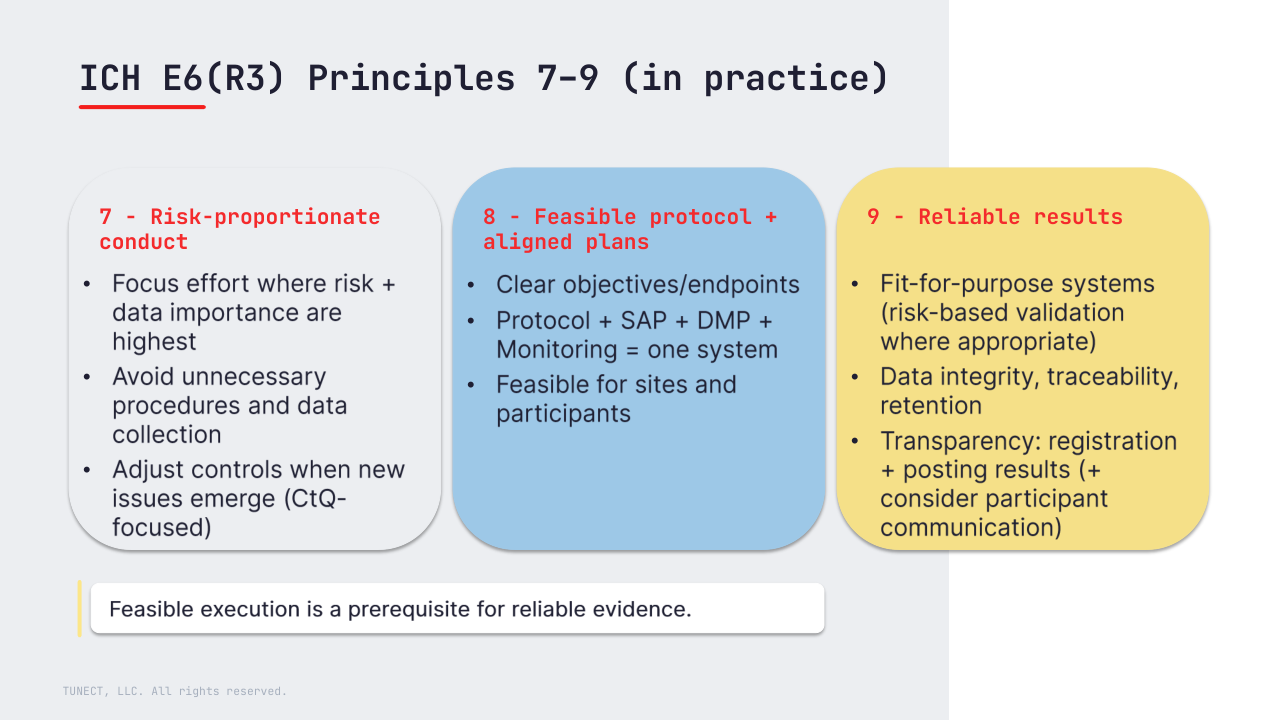
7) Risk-proportionate trial conduct (avoid unnecessary burden).
Processes should be proportionate to risks to participants (including risks beyond usual care where relevant) and to the importance of the data, while avoiding unnecessary procedures, complexity, and data collection.
R3 makes this an explicit principle: focus controls where they protect critical-to-quality (CtQ) factors, and adjust controls based on emerging issues and documented risk review.
8) Clear, concise, operationally feasible protocol.
A protocol should be scientifically sound and operationally feasible to run consistently. R3 reinforces that the protocol and its execution plans (SAP, DMP, monitoring) should work as one coherent system – consistent definitions, assumptions, and decision rules.
Feasibility is practical: what sites can deliver consistently and what participants can realistically complete.
9) Reliable results (fit-for-purpose systems + data integrity + transparency)
Trials should generate information that is fit for purpose and sufficient to support decision-making.
R3 strengthens expectations for fit-for-purpose computerized systems (with risk-based validation where appropriate), record integrity/traceability and retention, and transparency (timely registration and public posting of results; consider objective communication of results to participants).
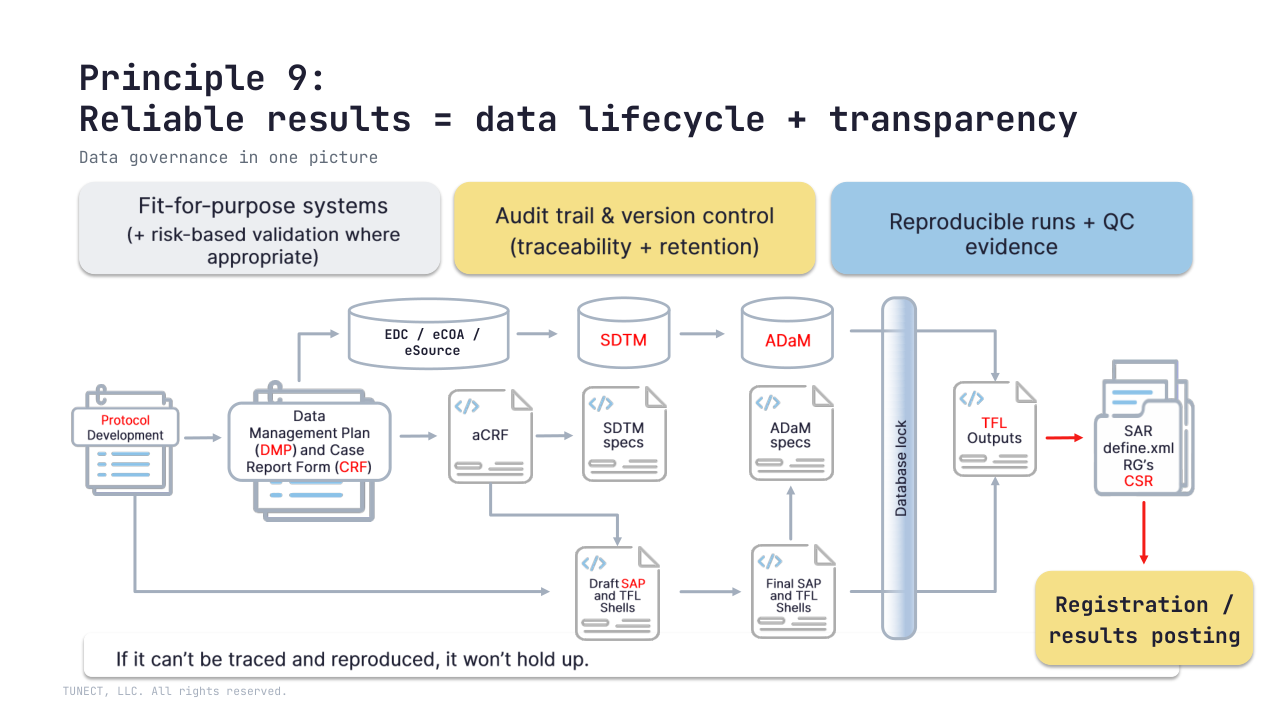
🧩 Biometrics/data lens: reliability shows up as an inspection-ready chain
Protocol → SAP/DMP → SDTM/ADaM → TFLs/CSR
with reproducible runs and QC evidence.
🔜 Next: Principles 10-11 – roles and responsibilities, and investigational products.
Question: where do you see the biggest pain today – operational burden, protocol feasibility, or end-to-end data traceability?