We’re closing our E6(R3) series with two principles that most often decide whether execution stays clean: role clarity and IP/blinding traceability.
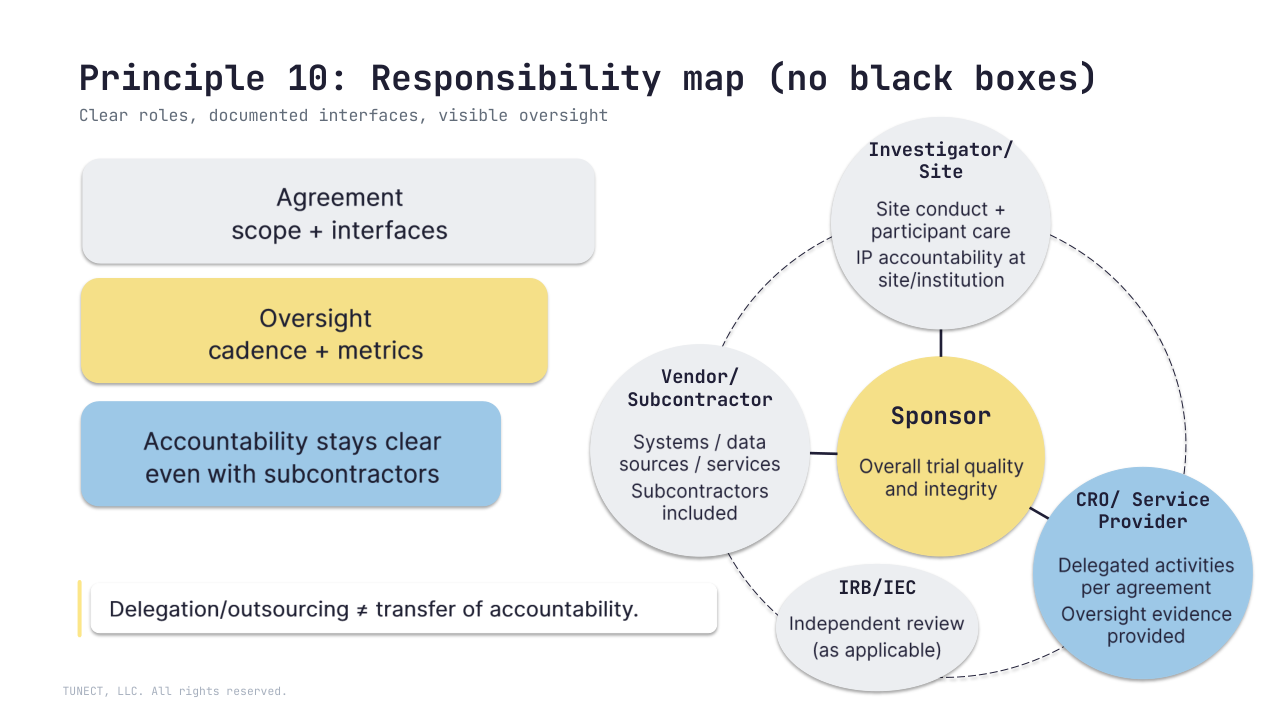
10) Roles and responsibilities (clarity + oversight)
Key roles, interfaces, and responsibilities should be clearly defined and documented appropriately across sponsors, investigators, service providers, vendors (and subcontractors).
What R3 makes explicit:
– Delegation/outsourcing ≠ transfer of accountability, oversight should remain in place
– Activities transferred to service providers should be documented in an agreement (scope, interfaces, oversight expectations)
– Overall responsibility for trial quality and integrity remains with the sponsor; investigators retain responsibility for site conduct and participant care, accountability stays clear across the chain
– And when vendors subcontract, responsibilities and oversight should stay explicit end-to-end
What “good” looks like: RACI / delegation log, vendor oversight plan, change-control triggers, and documented review/approval points; plus inspection-ready access to essential records (including those held by service providers).
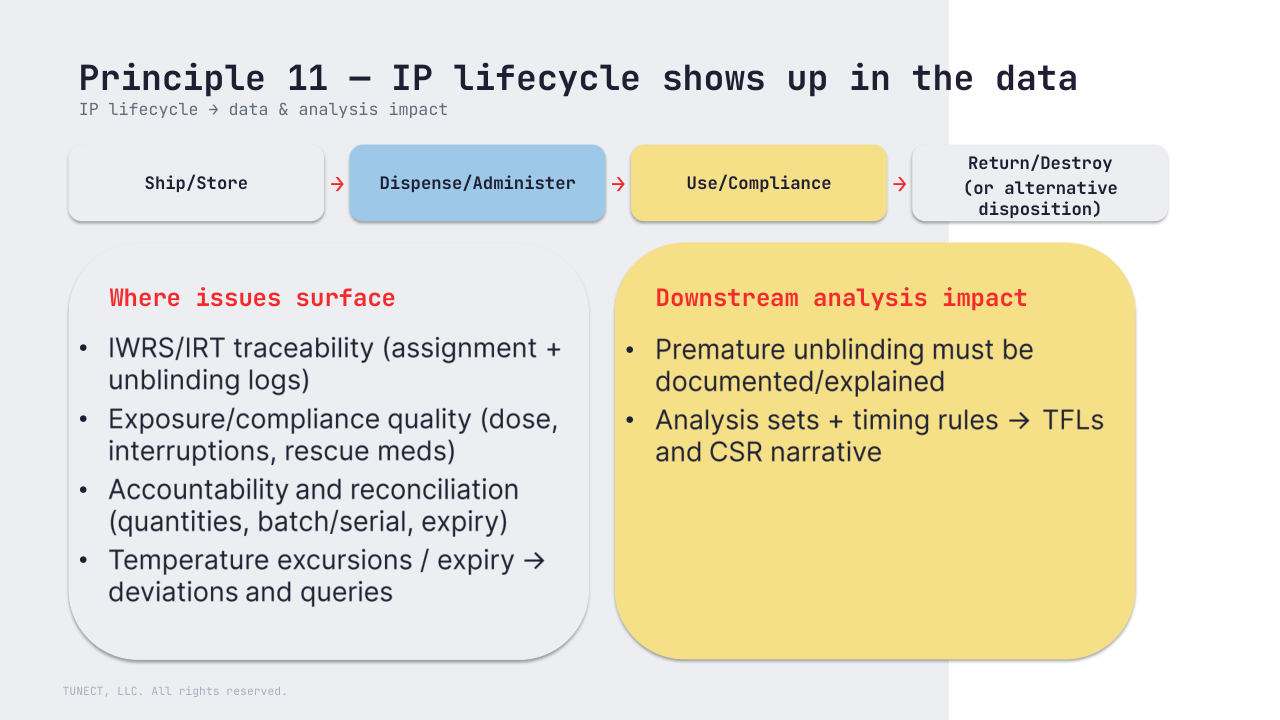
11) Investigational product (IP) oversight (lifecycle + blinding)
IP should be managed across its lifecycle in line with applicable requirements, the protocol, and blinding safeguards with documented accountability and disposition.
Accountability sits with the investigator/institution; the sponsor may facilitate distribution and tools.
Even for data-focused teams, this matters because it drives:
– Blinding/unblinding risk (roles, access, logging, including premature unblinding)
– Exposure/compliance and accountability record quality (dose, interruptions, rescue meds, reconciliation)
– Analysis sets and timing rules, and how they’re explained in TFLs and CSR narratives
– And it should be reflected upfront in the SAP (e.g., handling unblinding, treatment switching/rescue, and related sensitivity analyses)
Putting it together – the TUNECT approach
We focus on interfaces, evidence, and repeatability: transparent handoffs, reproducible runs, QC evidence, and a traceability chain Protocol -> SAP -> SDTM/ADaM -> TFLs/CSR, so narratives are supported by data, and review cycles are smoother.
Question: where do you see the biggest friction today – outsourced oversight, interface clarity, or IP/accountability data traceability?