In the last post we covered Principles 1-3 (ethics, consent, independent review). Now we move to the reliability backbone of trial evidence: science, people, and quality by design.
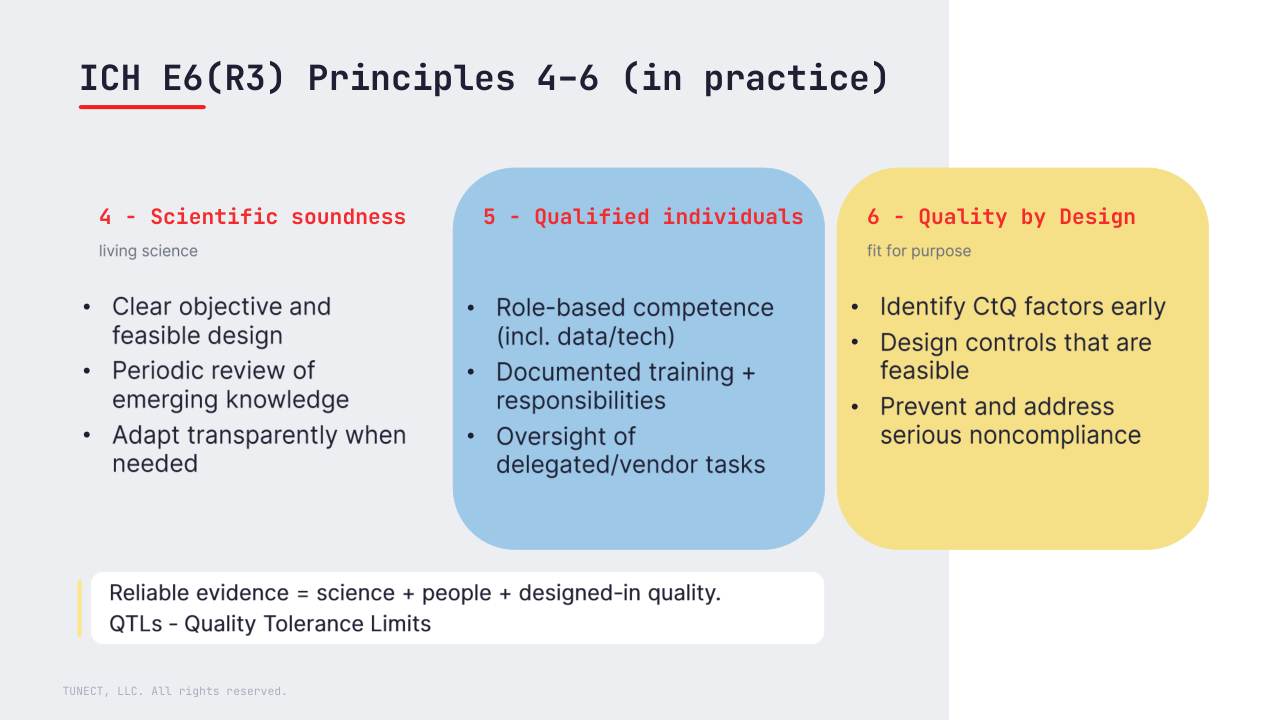
4) Scientifically sound trials (and periodic review)
Trials should be scientifically sound and aligned with current knowledge, with periodic review of emerging information and transparent adaptation when needed (with appropriate documentation and approvals where required).
🧩 Biostat lens: this is where the chain starts: objectives → endpoints → estimand/analysis strategy → data needed. If that link is weak, no amount of QC will “fix” interpretability later.
5) Qualified individuals
R3 makes the expectation explicit: people must be qualified for the tasks they actually perform (including data/technology roles, outsourced activities and service providers), with appropriate oversight.
In practice, this means role-based training, documented responsibilities, and clear evidence of review.
🧩 Think: training matrix, documented review steps, and vendor oversight evidence. R3 explicitly calls out roles like biostatisticians and data managers as part of the qualified team expectation.
6) Quality by Design (fitness for purpose)
R3 strengthens the idea that trial quality is fit for purpose – focused on what matters most for participant protection and reliable results.
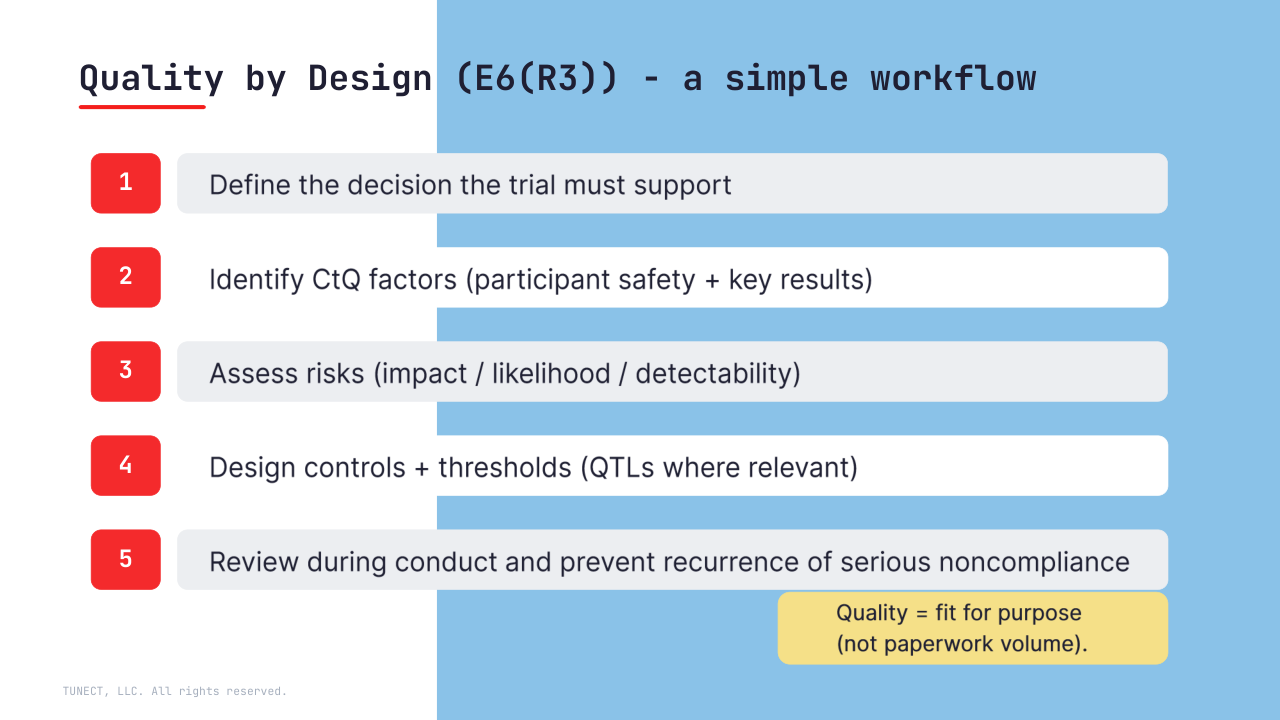
Quality by Design means identifying critical-to-quality (CtQ) factors early, designing feasible controls, and having strategies to prevent and address serious noncompliance – not only detect it after the fact.
ICHE6R3: “Where relevant, set pre-specified acceptable ranges (e.g., QTLs)…”
What this changes for biometrics/data teams (tomorrow morning):
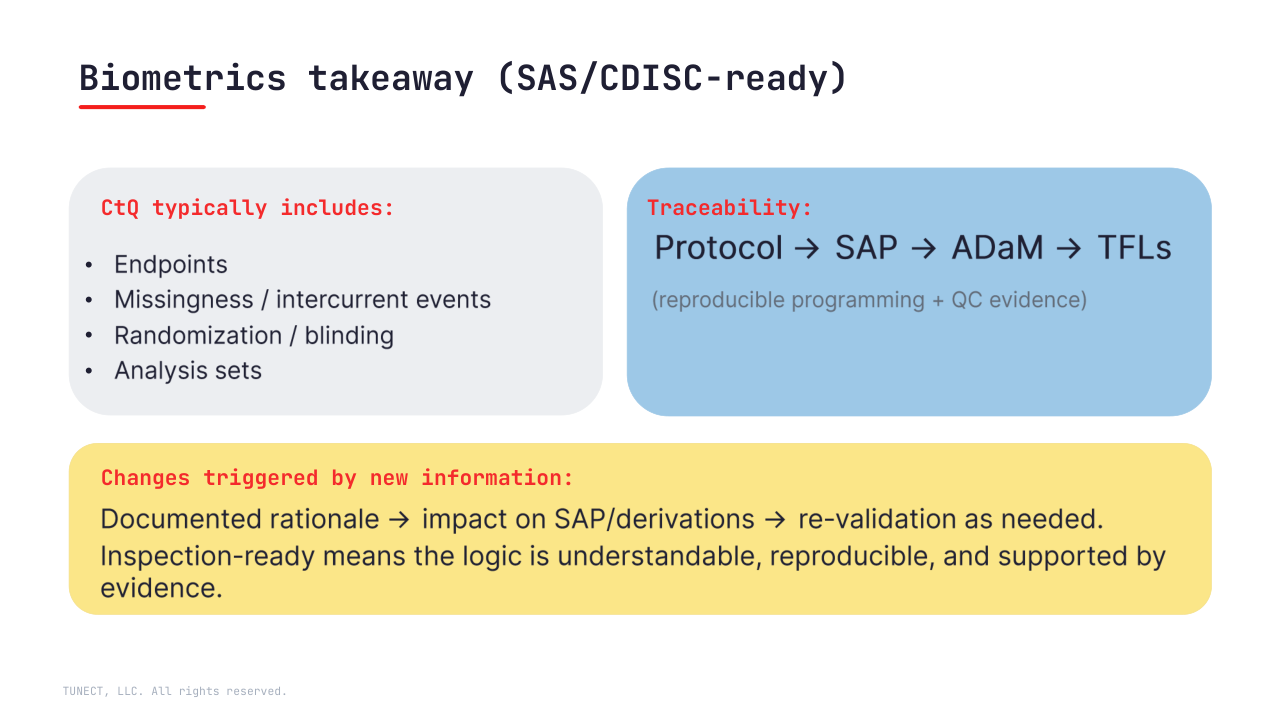
• Define CtQ around endpoints, key intercurrent events/missingness, randomization/blinding, and analysis sets
• Keep traceability inspection-ready: Protocol → SAP → ADaM → TFLs
(+ reproducible runs + QC evidence)
• Treat “emerging information” as a controlled change: documented rationale, impact assessment, and re-validation where needed
🔜 Next: Principles 7–9 – proportionality, protocol feasibility, and generating reliable results.
Question: where do you see the biggest gap today – scientific alignment, role qualification, or CtQ-focused quality planning?