What is a Functional Service Provider (FSP) in Clinical Research?
How does a CRO interact with a sponsor? What is project-based delivery, what is FSP...
GCP in Practice: Principles 10-11 of ICH E6(R3)
We’re closing our E6(R3) series with two principles that most often decide whether execution stays...
GCP in Practice: Principles 7-9 of ICH E6(R3)
Now we move from design principles to making execution feasible and ensuring the results are...
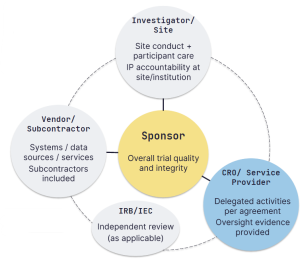
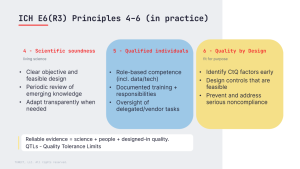
GCP in Practice: Principles 4-6 of ICH E6(R3)
In the last post we covered Principles 1-3 (ethics, consent, independent review). Now we move...
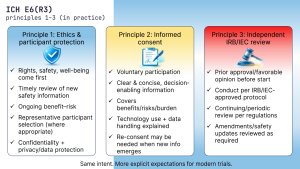
GCP in Practice: Principles 1–3 of ICH E6(R3)
We’re starting the series on the 11 principles of ICH E6(R3). E6(R3) consolidates and reframes...
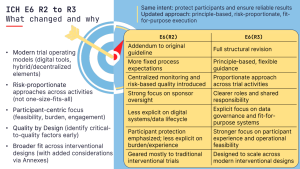
GCP in Practice: ICH E6(R2) → E6(R3) – why this update matters
The revision from ICH E6(R2) to E6(R3) was long overdue. Here’s what changed and what...
Data Protection Day: Privacy as a Foundation of Reliable Clinical Research
Data Protection Day (28 January): privacy in clinical research is not a separate legal topic...
October is Breast Cancer Awareness Month
To everyone living with breast cancer and to families, nurses, doctors, and site teams... we’re...
TUNECT CRO founders are heading to CPHI Frankfurt 2025
If you need senior, CDISC-native biometrics support with SDTM/ADaM/define.xml, Biostatistics, Data Management, Medical Writing (FSP...
TUNECT CRO at CRO Summit Europe
Thank you to everyone we met at CRO Summit Europe last week! It was superbly...